| Pharmaceutical Information |
| Drug Name |
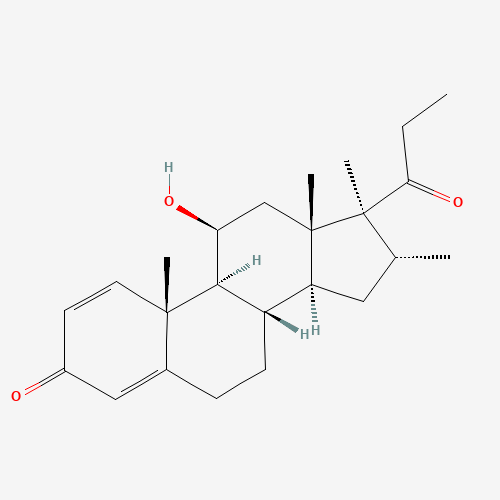
Rimexolone |
| Drug ID |
BADD_D01946 |
| Description |
Rimexolone is a 1% eye drop solution is a glucocorticoid steroid. It is used to treat inflammation in the eye. It is marketed as Vexol. |
| Indications and Usage |
For the treatment of postoperative inflammation following ocular surgery and in the treatment of anterior uveitis. |
| Marketing Status |
approved |
| ATC Code |
H02AB12; S01BA13 |
| DrugBank ID |
DB00896
|
| KEGG ID |
D05729
|
| MeSH ID |
C013769
|
| PubChem ID |
5311412
|
| TTD Drug ID |
D0D2TN
|
| NDC Product Code |
60870-0423 |
| UNII |
O7M2E4264D
|
| Synonyms |
rimexolone | 11 beta-hydroxy-16 alpha,17 alpha,21-trimethylpregna-1,4-diene-3,20-dione | Rimexel | Vexol | Org 6216 | Org-6216 |
|
| Chemical Information |
| Molecular Formula |
C24H34O3 |
| CAS Registry Number |
49697-38-3 |
| SMILES |
CCC(=O)C1(C(CC2C1(CC(C3C2CCC4=CC(=O)C=CC34C)O)C)C)C |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

