| Pharmaceutical Information |
| Drug Name |
Protriptyline hydrochloride |
| Drug ID |
BADD_D01873 |
| Description |
Protriptyline hydrochloride is a dibenzocycloheptene-derivative tricyclic antidepressant (TCA). TCAs are structurally similar to phenothiazines. They contain a tricyclic ring system with an alkyl amine substituent on the central ring. In non-depressed individuals, protriptyline does not affect mood or arousal, but may cause sedation. In depressed individuals, protriptyline exerts a positive effect on mood. TCAs are potent inhibitors of serotonin and norepinephrine reuptake. In addition, TCAs down-regulate cerebral cortical β-adrenergic receptors and sensitize post-synaptic serotonergic receptors with chronic use. The antidepressant effects of TCAs are thought to be due to an overall increase in serotonergic neurotransmission. TCAs also block histamine H1 receptors, alpha1-adrenergic receptors and muscarinic receptors, which accounts for their sedative, hypotensive and anticholinergic effects (e.g. blurred vision, dry mouth, constipation, urinary retention), respectively. See toxicity section below for a complete listing of side effects. Protriptyline may be used for the treatment of depression. |
| Indications and Usage |
For the treatment of depression. |
| Marketing Status |
approved |
| ATC Code |
N06AA11 |
| DrugBank ID |
DB00344
|
| KEGG ID |
D00484
|
| MeSH ID |
D011530
|
| PubChem ID |
6603149
|
| TTD Drug ID |
D00MYQ
|
| NDC Product Code |
0054-0210; 46438-0056; 0054-0211; 42794-004; 42806-096; 46438-0066; 42806-097; 17337-0539; 42794-007 |
| UNII |
44665V00O8
|
| Synonyms |
Protriptyline | Vivactil | Protriptyline Hydrochloride | Hydrochloride, Protriptyline |
|
| Chemical Information |
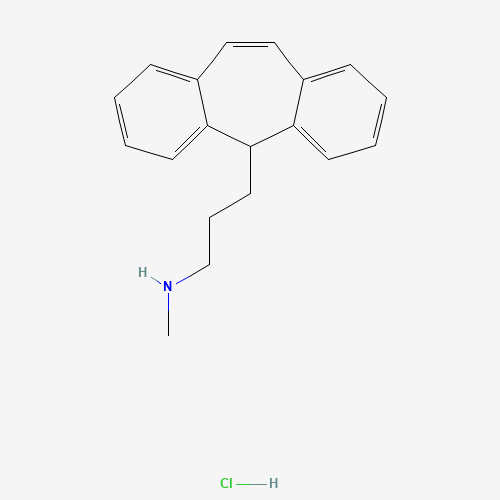
| Molecular Formula |
C19H22ClN |
| CAS Registry Number |
1225-55-4 |
| SMILES |
CNCCCC1C2=CC=CC=C2C=CC3=CC=CC=C13.Cl |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Bone marrow failure | 01.03.03.005 | - | - | |
|
|
|

