| Pharmaceutical Information |
| Drug Name |
Pirbuterol |
| Drug ID |
BADD_D01781 |
| Description |
Pirbuterol is a beta-2 adrenergic bronchodilator. In vitro studies and in vivo pharmacologic studies have demonstrated that pirbuterol has a preferential effect on beta-2 Adrenergic receptors compared with isoproterenol. While it is recognized that beta-2 adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that there is a population of beta-2 receptors in the human heart, existing in a concentration between 10-50%. The precise function of these receptors has not been established.
The pharmacologic effects of beta adrenergic agonist drugs, including pirbuterol, are at least in proof attributable to stimulation through beta adrenergic receptors of intracellular adenyl cyclase, the enzyme which catalyzes the conversion of adenosine triphosphate (AlP) to cyclic-3† ,5†-adenosine monophosphate (c-AMP). Increased c-AMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells. |
| Indications and Usage |
For the prevention and reversal of bronchospasm in patients 12 years of age and older with reversible bronchospasm including asthma. |
| Marketing Status |
approved |
| ATC Code |
R03AC08; R03CC07 |
| DrugBank ID |
DB01291
|
| KEGG ID |
D08387
|
| MeSH ID |
C009118
|
| PubChem ID |
4845
|
| TTD Drug ID |
D0SS4P
|
| NDC Product Code |
Not Available |
| UNII |
OG645J8RVW
|
| Synonyms |
pirbuterol | pyrbuterol | CP-24,314-1 | Maxair | pirbuterol dihydrochloride | 2-hydroxymethyl-3-hydroxy-6-(1-hydroxy-2-tert-butylamino ethyl)pyridine, dihydrochloride | pirbuterol sulfate | CP 24315-1 | CP-24315-1 | pirbuterol acetate | pirbuterol acetate salt |
|
| Chemical Information |
| Molecular Formula |
C12H20N2O3 |
| CAS Registry Number |
38677-81-5 |
| SMILES |
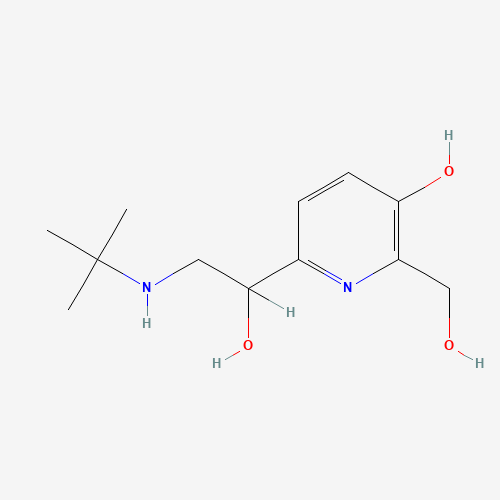
CC(C)(C)NCC(C1=NC(=C(C=C1)O)CO)O |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

