| Pharmaceutical Information |
| Drug Name |
Ofev |
| Drug ID |
BADD_D01597 |
| Description |
Nintedanib is a small molecule kinase inhibitor used in the treatment of pulmonary fibrosis, systemic sclerosis-associated interstitial lung disease, and non-small cell lung cancer (NSCLC).[L8453,L8459] It was first approved for use in the United States in 2014.[L8453] Within the spectrum of idiopathic pulmonary fibrosis treatment options, nintedanib is currently one of only two disease-modifying therapies available and indicated for the condition (the other being [Pirfenidone]) and as such is used as a first-line treatment following diagnosis to slow down the progressive loss of lung function.[A185237] As a chemotherapeutic agent for NSCLC, nintedanib, in combination with [Docetaxel], is reserved for patients who have tried and failed first-line chemotherapeutic options.[L8459] |
| Indications and Usage |
In the US, nintedanib is indicated for the treatment of idiopathic pulmonary fibrosis (IPF)[L8453] and to slow declining pulmonary function in patients with systemic sclerosis-associated interstitial lung disease.[L8462] In the EU, nintedanib is indicated in combination with docetaxel for the treatment of adult patients with metastatic, locally advanced, or locally recurrent non-small cell lung cancer of adenocarcinoma histology who have already tried first-line therapy.[L8459] |
| Marketing Status |
approved |
| ATC Code |
L01EX09 |
| DrugBank ID |
DB09079
|
| KEGG ID |
D10396; D10481
|
| MeSH ID |
C530716
|
| PubChem ID |
135423438
|
| TTD Drug ID |
D09HNV
|
| NDC Product Code |
55361-0020; 66499-0061; 0597-0143; 0597-0145; 51869-0040; 55361-0021 |
| UNII |
42F62RTZ4G
|
| Synonyms |
nintedanib | Nintedanib esylate | Ofev | Vargatef | BIBF 1120 | BIBF1120 | BIBF-1120 |
|
| Chemical Information |
| Molecular Formula |
C31H33N5O4 |
| CAS Registry Number |
656247-17-5 |
| SMILES |
CN1CCN(CC1)CC(=O)N(C)C2=CC=C(C=C2)N=C(C3=CC=CC=C3)C4=C(NC5=C4C=CC(=C5)C(=O)OC)O |
| Chemical Structure |
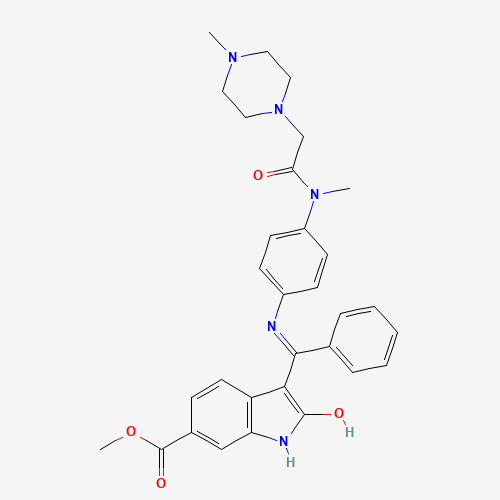
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

