| Pharmaceutical Information |
| Drug Name |
Magnevist |
| Drug ID |
BADD_D01345 |
| Description |
A complex of gadolinium with a chelating agent, diethylenetriamine penta-acetic acid (DTPA see pentetic acid), that is given to enhance the image in cranial and spinal MRIs. (From Martindale, The Extra Pharmacopoeia, 30th ed, p706) |
| Indications and Usage |
For use with magnetic resonance imaging (MRI) in adults, and pediatric patients (2 years of age and older) to visualize lesions with abnormal vascularity in the brain (intracranial lesions), spine and associated tissues as well as lesions with abnormal vascularity in the head and neck. Also used to facilitate the visualization of lesions with abnormal vascularity in the body (excluding the heart). |
| Marketing Status |
approved |
| ATC Code |
V08CA01 |
| DrugBank ID |
DB00789
|
| KEGG ID |
D01707
|
| MeSH ID |
D019786
|
| PubChem ID |
55466
|
| TTD Drug ID |
D0RQ2W
|
| NDC Product Code |
Not Available |
| UNII |
RH248G8V27
|
| Synonyms |
Gadolinium DTPA | DTPA, Gadolinium | Gadolinium Diethylenetriaminepenta-acetic Acid | Diethylenetriaminepenta-acetic Acid, Gadolinium | Gadolinium Diethylenetriaminepenta acetic Acid | Gadopentetic Acid | Gd-DTPA | Gd DTPA | Gadolinium DTPA Disodium Salt | Magnograf | Magnograf Enteral | Gadopentetate Dimeglumine | Dimeglumine, Gadopentetate | Gadolinium DTPA Dimeglumine Salt | Gadolinium DTPA Dimeglumine | Dimeglumine, Gadolinium DTPA | Magnevist | Magnevist Enteral |
|
| Chemical Information |
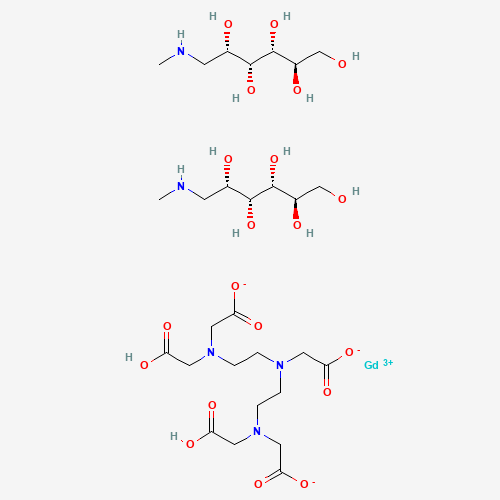
| Molecular Formula |
C28H54GdN5O20 |
| CAS Registry Number |
80529-93-7 |
| SMILES |
CNCC(C(C(C(CO)O)O)O)O.CNCC(C(C(C(CO)O)O)O)O.C(CN(CC(=O)O)CC(=O)[O-])N(CCN(CC(=O)
O)CC(=O)[O-])CC(=O)[O-].[Gd+3] |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

