| Pharmaceutical Information |
| Drug Name |
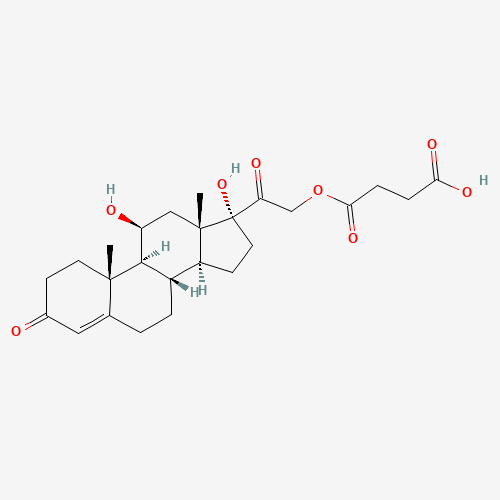
Hydrocortisone succinate |
| Drug ID |
BADD_D02523 |
| Description |
Not Available |
| Indications and Usage |
Not Available |
| Marketing Status |
Not Available |
| ATC Code |
A01AC03; A07EA02; C05AA01; D07AA02; D07XA01; H02AB09; S01BA02; S01CB03; S02BA01 |
| DrugBank ID |
DB14545
|
| KEGG ID |
D01442
|
| MeSH ID |
C007133
|
| PubChem ID |
16623
|
| TTD Drug ID |
Not Available
|
| NDC Product Code |
Not Available |
| Synonyms |
hydrocortisone hemisuccinate | cortisol-21-(hydrogen succinate) | cortisol hemisuccinate | cortisol succinate, sodium salt | hydrocortisone 21-sodium succinate | Sopolkort | hydrocortisone succinate | pregn-4-ene-3,20-dione, 21-(3-carboxy-1-oxopropoxy)-11,17-dihydroxy-, (11beta)- | hydrocortisone hemisuccinate anhydrous | cortisol succinate | Sopolcort H | A-hyrocort | Solu-Cortef |
|
| Chemical Information |
| Molecular Formula |
C25H34O8 |
| CAS Registry Number |
2203-97-6 |
| SMILES |
CC12CCC(=O)C=C1CCC3C2C(CC4(C3CCC4(C(=O)COC(=O)CCC(=O)O)O)C)O |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
|

