| Pharmaceutical Information |
| Drug Name |
Valbenazine |
| Drug ID |
BADD_D02491 |
| Description |
Valbenazine (development name NBI-98854) has been used in trials studying the treatment and basic science of Tourette Syndrome and Tardive Dyskinesia. In April, 2017, valbenazine was approved by the FDA (as Ingrezza) as the first and only approved treatment for adults with Tardive Dyskinesia (TD). |
| Indications and Usage |
For the treatment of tardive dyskinesia in adults [FDA Label]. |
| Marketing Status |
Not Available |
| ATC Code |
N07XX13 |
| DrugBank ID |
DB11915
|
| KEGG ID |
D10675
|
| MeSH ID |
C000603978
|
| PubChem ID |
24795069
|
| TTD Drug ID |
D63WMQ
|
| NDC Product Code |
70370-1080; 47049-008; 70370-1060; 66406-0314; 47049-021; 70370-2040; 66406-0274; 70370-2048; 47049-009 |
| Synonyms |
valbenazine | (2R,3R,11BR)-9,10-dimethoxy-3-(2-methylpropyl)-1,3,4,6,7,11b-hexahydro-2H- benzo(a)quinolizin-2-yl l-valinate | NBI-98854 | Ingrezza | valine 1,3,4,6,7,11b-hexahydro-9,10-dimethoxy-3-(2-methylpropyl)-2H-benzo(a)quinolizin-2-yl ester |
|
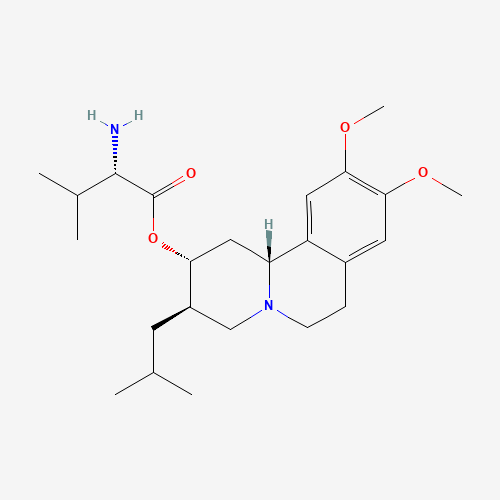
| Chemical Information |
| Molecular Formula |
C24H38N2O4 |
| CAS Registry Number |
1025504-45-3 |
| SMILES |
CC(C)CC1CN2CCC3=CC(=C(C=C3C2CC1OC(=O)C(C(C)C)N)OC)OC |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Tachyphrenia | 19.10.03.010; 17.03.03.007 | 0.002614% | | Not Available |
|
|
|

