| Pharmaceutical Information |
| Drug Name |
Niraparib |
| Drug ID |
BADD_D02486 |
| Description |
Niraparib is an orally active PARP inhibitor developed by Tesaro to treat ovarian cancer. FDA approval on March 2017. |
| Indications and Usage |
Niraparib is indicated for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in complete or partial response to platinum-based chemotherapy |
| Marketing Status |
Not Available |
| ATC Code |
L01XK02 |
| DrugBank ID |
DB11793
|
| KEGG ID |
D10140
|
| MeSH ID |
C545685
|
| PubChem ID |
24958200
|
| TTD Drug ID |
D00BMF
|
| NDC Product Code |
71794-103; 69656-103; 61876-0723 |
| Synonyms |
niraparib | 2-(4-(piperidin-3-yl)phenyl)-2H-indazole-7-carboxamide | niraparib hydrochloride | Zejula | MK 4827 | MK4827 | MK-4827 |
|
| Chemical Information |
| Molecular Formula |
C19H20N4O |
| CAS Registry Number |
1038915-60-4 |
| SMILES |
C1CC(CNC1)C2=CC=C(C=C2)N3C=C4C=CC=C(C4=N3)C(=O)N |
| Chemical Structure |
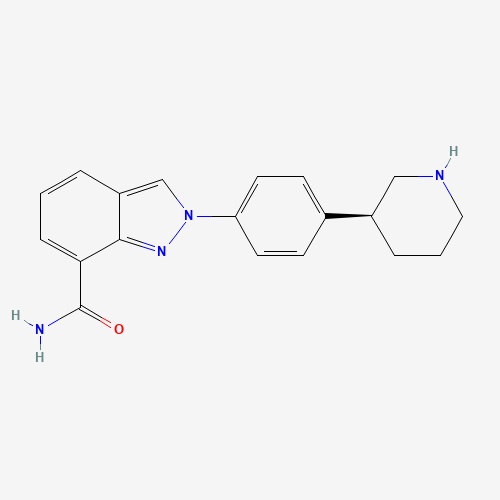
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Scan abnormal | 13.18.03.005 | 0.000533% | | Not Available | | Tongue discomfort | 07.14.02.019 | 0.000799% | | Not Available |
|
|
|

