| Pharmaceutical Information |
| Drug Name |
Trisodium citrate dihydrate |
| Drug ID |
BADD_D02300 |
| Description |
Sodium citrate is the sodium salt of citric acid. It is white, crystalline powder or white, granular crystals, slightly deliquescent in moist air, freely soluble in water,
practically insoluble in alcohol. Like citric acid, it has a sour taste.
From the medical point of view, it is used as alkalinizing agent. It works by neutralizing excess acid in the blood and urine. It has been indicated for the treatment of metabolic acidosis. |
| Indications and Usage |
Used as an anticoagulant during plasmophoresis as well as a neutralizing agent in the treatment of upset stomach and acidic urine [FDA Label] [L788] [L789]. |
| Marketing Status |
approved; investigational |
| ATC Code |
B05CB02 |
| DrugBank ID |
DB09154
|
| KEGG ID |
D01781
|
| MeSH ID |
D000077559
|
| PubChem ID |
71474
|
| TTD Drug ID |
D05KTE
|
| NDC Product Code |
23731-6030; 57826-420; 76297-006; 14537-883; 60929-832; 14537-881; 81839-782 |
| UNII |
B22547B95K
|
| Synonyms |
Sodium Citrate | Trisodium Citrate Dihydrate | Sodium Citrate Dihydrate | Monosodium Citrate | Sodium Citrate Monobasic | Sodium Citrate, Anhydrous | Anhydrous Sodium Citrate | Citra pH |
|
| Chemical Information |
| Molecular Formula |
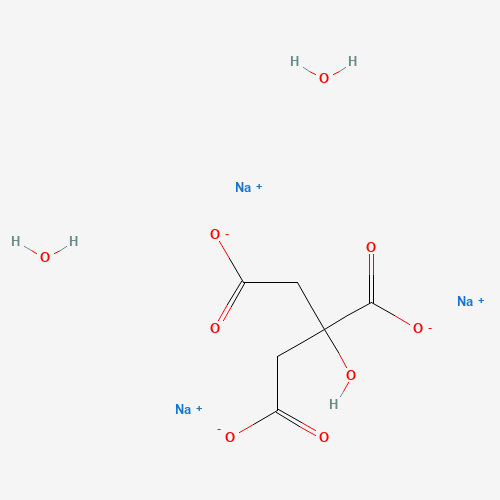
C6H9Na3O9 |
| CAS Registry Number |
6132-04-3 |
| SMILES |
C(C(=O)[O-])C(CC(=O)[O-])(C(=O)[O-])O.O.O.[Na+].[Na+].[Na+] |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Citrate toxicity | 14.11.01.026; 12.03.01.043 | - | - | Not Available |
|
The 1th Page
1
Total 1 Pages
|
|

