| Pharmaceutical Information |
| Drug Name |
Regorafenib |
| Drug ID |
BADD_D01922 |
| Description |
Regorafenib is an orally-administered inhibitor of multiple kinases. It is used for the treatment of metastatic colorectal cancer, advanced gastrointestinal stromal tumours, and hepatocellular carcinoma. FDA approved on September 27, 2012. Approved use of Regorafenib was expanded to treat Hepatocellular Carcinoma in April 2017. |
| Indications and Usage |
Regorafenib is indicated for the treatment of patients with metastatic colorectal cancer (CRC) who have been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy, an anti-VEGF therapy, and, if KRAS wild type, an anti-EGFR therapy. Regorafenib is also indicated for the treatment of patients with locally advanced, unresectable or metastatic gastrointestinal stromal tumor (GIST) who have been previously treated with imatinib mesylate and sunitinib malate. |
| Marketing Status |
Prescription |
| ATC Code |
L01EX05 |
| DrugBank ID |
DB08896
|
| KEGG ID |
D10138
|
| MeSH ID |
C559147
|
| PubChem ID |
11167602
|
| TTD Drug ID |
D09GDD
|
| NDC Product Code |
12527-0171; 50419-171; 54893-0033; 63415-0522 |
| Synonyms |
regorafenib | 4-(4-(((4-chloro-3-(trifluoromethyl)phenyl)carbamoyl)amino)-3-fluorophenoxy)-n-methylpyridine-2-carboxamide | Stivarga | BAY 73-4506 | BAY73-4506 | BAY-73-4506 |
|
| Chemical Information |
| Molecular Formula |
C21H15ClF4N4O3 |
| CAS Registry Number |
755037-03-7 |
| SMILES |
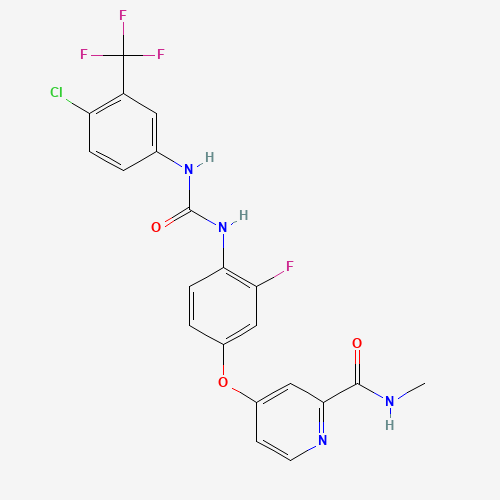
CNC(=O)C1=NC=CC(=C1)OC2=CC(=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F)F |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
|

