| Pharmaceutical Information |
| Drug Name |
Pyrimethamine |
| Drug ID |
BADD_D01886 |
| Description |
One of the folic acid antagonists that is used as an antimalarial or with a sulfonamide to treat toxoplasmosis. |
| Indications and Usage |
For the treatment of toxoplasmosis and acute malaria; For the prevention of malaria in areas non-resistant to pyrimethamine |
| Marketing Status |
Prescription |
| ATC Code |
P01BD01 |
| DrugBank ID |
DB00205
|
| KEGG ID |
D00488
|
| MeSH ID |
D011739
|
| PubChem ID |
4993
|
| TTD Drug ID |
D0C0SK
|
| NDC Product Code |
51927-0103; 0480-3720; 43598-672; 72647-330; 60592-244; 57451-1183; 66651-922; 64677-0006; 47781-925; 51927-3122; 67763-114; 68057-001; 63804-5300; 51927-0237; 69413-330; 48102-026; 59651-564; 71052-215; 40074-0008; 71554-020 |
| Synonyms |
Pyrimethamine | Malocide | Tindurine | Chloridin | Daraprim |
|
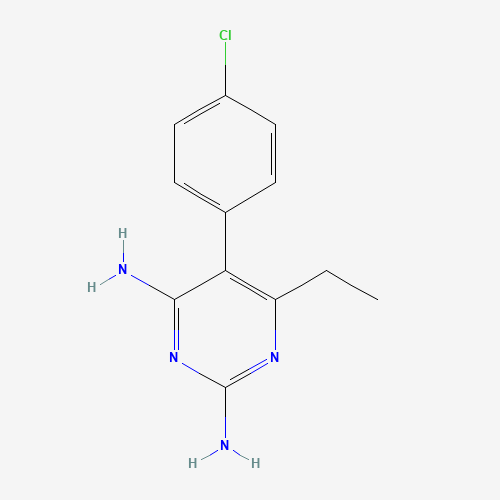
| Chemical Information |
| Molecular Formula |
C12H13ClN4 |
| CAS Registry Number |
58-14-0 |
| SMILES |
CCC1=C(C(=NC(=N1)N)N)C2=CC=C(C=C2)Cl |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Kidney congestion | 20.02.03.015; 02.05.04.017 | 0.000122% | | Not Available |
|
|
|

