| Pharmaceutical Information |
| Drug Name |
Primaquine |
| Drug ID |
BADD_D01838 |
| Description |
An aminoquinoline that is given by mouth to produce a radical cure and prevent relapse of vivax and ovale malarias following treatment with a blood schizontocide. It has also been used to prevent transmission of falciparum malaria by those returning to areas where there is a potential for re-introduction of malaria. Adverse effects include anemias and GI disturbances. (From Martindale, The Extra Pharmacopeia, 30th ed, p404) |
| Indications and Usage |
For the treatment of malaria. |
| Marketing Status |
approved |
| ATC Code |
P01BA03 |
| DrugBank ID |
DB01087
|
| KEGG ID |
D02126; D08420
|
| MeSH ID |
D011319
|
| PubChem ID |
4908
|
| TTD Drug ID |
D0T1LK
|
| NDC Product Code |
Not Available |
| UNII |
MVR3634GX1
|
| Synonyms |
Primaquine | Primaquine Phosphate | Phosphate, Primaquine | Primacin | Primaquine Diphosphate | Diphosphate, Primaquine |
|
| Chemical Information |
| Molecular Formula |
C15H21N3O |
| CAS Registry Number |
90-34-6 |
| SMILES |
CC(CCCN)NC1=C2C(=CC(=C1)OC)C=CC=N2 |
| Chemical Structure |
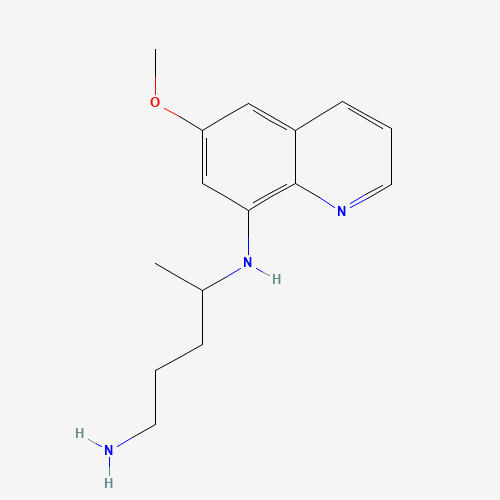
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

