| Pharmaceutical Information |
| Drug Name |
Porfimer sodium |
| Drug ID |
BADD_D01803 |
| Description |
The purified component of hematoporphyrin derivative, it consists of a mixture of oligomeric porphyrins. It is used in photodynamic therapy (hematoporphyrin photoradiation); to treat malignant lesions with visible light and experimentally as an antiviral agent. It is the first drug to be approved in the use of photodynamic therapy in the United States. |
| Indications and Usage |
Indicated in the treatment of esophageal cancer. |
| Marketing Status |
Prescription |
| ATC Code |
L01XD01 |
| DrugBank ID |
DB00707
|
| KEGG ID |
D03327
|
| MeSH ID |
D017323
|
| PubChem ID |
135577896
|
| TTD Drug ID |
D02JDL
|
| NDC Product Code |
76128-155 |
| Synonyms |
Dihematoporphyrin Ether | Ether, Dihematoporphyrin | Dihematoporphyrin Ester | Porfimer | DHP Ether | Porfimer Sodium | Photofrin | Photofrin II |
|
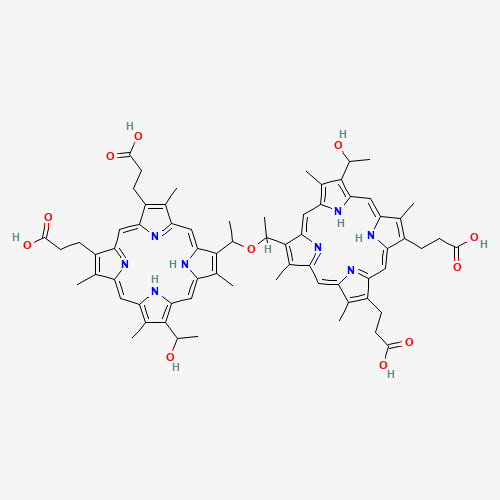
| Chemical Information |
| Molecular Formula |
C68H74N8O11 |
| CAS Registry Number |
87806-31-3 |
| SMILES |
CC1=C(C2=CC3=NC(=CC4=NC(=CC5=C(C(=C(N5)C=C1N2)C(C)O)C)C(=C4C)C(C)OC(C)C6=C(C7=CC
8=C(C(=C(N8)C=C9C(=C(C(=N9)C=C1C(=C(C(=N1)C=C6N7)C)CCC(=O)O)CCC(=O)O)C)C)C(C)O)C
)C(=C3CCC(=O)O)C)CCC(=O)O |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Aorto-oesophageal fistula | 24.07.02.041; 07.11.05.006 | 0.000347% | | Not Available |
|
|
|

