| Pharmaceutical Information |
| Drug Name |
Pazopanib |
| Drug ID |
BADD_D01687 |
| Description |
Pazopanib is a small molecule inhibitor of multiple protein tyrosine kinases with potential antineoplastic activity. It is developed by GlaxoSmithKline and was FDA approved on October 19, 2009. |
| Indications and Usage |
Treatment of advanced renal cell cancer and advanced soft tissue sarcoma (in patients previously treated with chemotherapy) |
| Marketing Status |
Prescription; Discontinued |
| ATC Code |
L01EX03 |
| DrugBank ID |
DB06589
|
| KEGG ID |
D05380
|
| MeSH ID |
C516667
|
| PubChem ID |
10113978
|
| TTD Drug ID |
Not Available
|
| NDC Product Code |
Not Available |
| Synonyms |
pazopanib | GW 786034B | GW786034B | GW-786034B | GW 780604 | GW780604 | GW-780604 | Votrient |
|
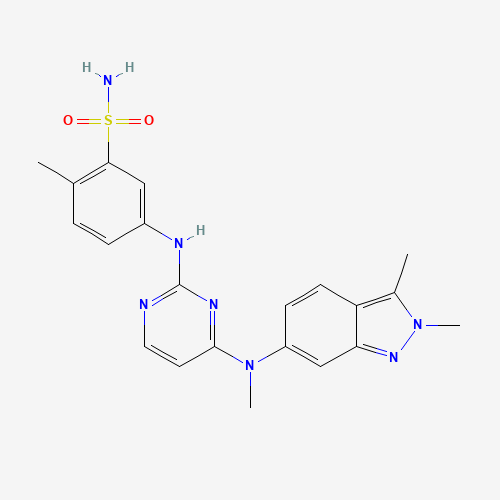
| Chemical Information |
| Molecular Formula |
C21H23N7O2S |
| CAS Registry Number |
444731-52-6 |
| SMILES |
CC1=C(C=C(C=C1)NC2=NC=CC(=N2)N(C)C3=CC4=NN(C(=C4C=C3)C)C)S(=O)(=O)N |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
|

