| Pharmaceutical Information |
| Drug Name |
Lisinopril |
| Drug ID |
BADD_D01300 |
| Description |
Lisinopril is an angiotensin converting enzyme inhibitor (ACEI) used to treat hypertension, heart failure, and myocardial infarction.[L8384,L8387,L8390] Lisinopril and [captopril] are the only ACEIs that are not prodrugs.[A184853] It functions by inhibition of angiotensin converting enzyme as well as the renin angiotensin aldosterone system.[A184781,A184808,A184817] ACEIs are commonly used as a first line therapy in the treatment of hypertension, along with thiazide diuretics or beta blockers.[A184844]
Lisinopril was granted FDA approval on 29 December 1987.[L8384] |
| Indications and Usage |
Lisinopril is indicated for the treatment of acute myocardial infarction, hypertension in patients ≥6 years, and as an adjunct therapy for heart failure.[L8384,L8387] A combination product with hydrochlorothiazide is indicated for the treatment of hypertension.[L8390] |
| Marketing Status |
approved; investigational |
| ATC Code |
C09AA03 |
| DrugBank ID |
DB00722
|
| KEGG ID |
D00362; D08131
|
| MeSH ID |
D017706
|
| PubChem ID |
5362119
|
| TTD Drug ID |
D07HGR
|
| NDC Product Code |
64220-154; 65862-037; 16571-797; 16571-798; 42708-031; 42708-033; 43353-270; 43353-271; 43547-417; 50090-2228; 50090-4678; 51655-707; 53002-1225; 55154-2128; 60687-325; 60760-359; 61919-246; 62135-641; 63187-099; 63629-8823; 65862-039; 65862-040; 65862-042; 68001-336; 68071-5143; 68084-196; 68645-551; 68645-587; 68645-613; 70518-0117; 70518-2600; 71335-0176; 71335-1380; 71610-612; 0615-8254; 76282-419; 76282-729; 0591-0405; 0591-0409; 0904-6797; 0904-7200; 57297-511; 63827-1012; 16729-376; 16729-379; 43353-113; 43353-993; 43547-351; 43547-355; 50090-3763; 51655-146; 55154-8096; 55289-884; 60687-333; 61919-746; 62135-642; 63187-237; 63187-442; 65862-038; 68001-334; 68071-3220; 68180-982; 68645-608; 68645-610; 68788-6407; 70518-1790; 70518-1906; 0591-0406; 71335-0354; 71610-588; 0615-8253; 72865-208; 72865-209; 76282-422; 76282-731; 76282-733; 82009-063; 65862-041; 65977-0009; 23155-714; 31722-172; 42708-142; 43353-301; 50090-3765; 50090-3774; 50090-4725; 50090-4885; 50436-0352; 50436-0354; 51655-399; 51655-729; 51655-999; 58118-1980; 60687-678; 60760-380; 62135-644; 63187-775; 63629-5247; 67296-1202; 67296-1839; 68180-517; 68180-981; 68645-550; 68645-555; 69117-0037; 70882-104; 71205-471; 71335-1295; 71610-624; 0615-8255; 61919-909; 62135-645; 63187-257; 63187-821; 63629-8736; 68001-335; 68180-513; 68645-593; 68788-7012; 68788-7334; 69117-0036; 70518-0544; 70882-103; 70934-155; 71205-180; 71205-742; 71335-0015; 71610-638; 0615-8405; 72865-210; 82009-065; 82982-066; 14593-944; 17404-0011; 16571-794; 16571-795; 42708-154; 43547-418; 43547-419; 50090-0808; 52427-441; 58118-1981; 60760-360; 61919-721; 62135-643; 63187-534; 63629-1761; 67296-1452; 68001-337; 68071-3123; 68645-552; 68645-553; 68645-609; 68788-6820; 68788-6920; 69117-0038; 69117-1003; 70518-1152; 70882-110; 70934-338; 70934-522; 70934-919; 71205-071; 76282-728; 76282-730; 82009-066; 0591-0885; 16571-796; 16729-377; 43063-480; 43063-810; 43063-812; 43547-353; 51655-486; 52427-438; 53002-1123; 60687-646; 60687-667; 63187-825; 68001-332; 68001-486; 68071-3409; 68071-5190; 68180-512; 68180-979; 68180-980; 68645-554; 68645-611; 68788-8351; 70518-0468; 70934-171; 71205-626; 0615-8252; 0904-6800; 67651-0225; 31722-179; 31722-180; 42708-176; 43063-769; 43547-352; 43547-354; 43547-356; 50090-2989; 50090-3175; 51655-995; 52652-3001; 53002-1178; 53002-1463; 60760-358; 60760-390; 60760-438; 61919-717; 61919-736; 63187-098; 63187-780; 68001-333; 68071-2224; 68071-3411; 68084-765; 70518-2561; 70518-2655; 0591-0407; 72789-093; 72789-101; 72865-211; 76282-420; 76282-421; 82009-064; 64220-142; 16729-380; 23155-710; 23155-711; 23155-712; 43063-786; 43063-800; 43063-811; 43063-813; 43353-297; 43353-643; 43353-662; 43353-793; 43547-415; 50090-6303; 51655-335; 51655-411; 52427-442; 62135-640; 63187-722; 68071-3064; 68788-8310; 69117-1001; 71335-0075; 71610-312; 71610-406; 71610-536; 72865-212; 76282-418; 0904-6798; 0904-6799; 65730-1301; 42708-096; 42708-165; 50090-2431; 50090-4887; 52427-443; 55154-4992; 55154-8078; 55700-480; 60760-357; 60760-455; 68645-532; 70518-1156; 70518-1316; 70518-3166; 0591-0408; 76282-417; 16571-799; 16729-375; 31722-176; 31722-178; 42708-032; 43063-074; 43353-009; 43353-298; 43547-414; 43547-416; 50090-5120; 50436-0353; 51655-994; 52427-440; 53002-2123; 55154-2329; 55700-479; 55700-547; 60687-656; 67296-1154; 68645-612; 68788-7666; 69117-1002; 71335-0498; 71335-0536; 72865-213; 76282-732; 82009-062; 16729-378; 23155-713; 23155-715; 31722-177; 42708-094; 43063-814; 43063-815; 52427-439 |
| UNII |
E7199S1YWR
|
| Synonyms |
Lisinopril | Lysinopril | Lisinopril Sulfate (1:2) | Prinivil | Zestril | Lisinopril Maleate (1:1) | MK-521 |
|
| Chemical Information |
| Molecular Formula |
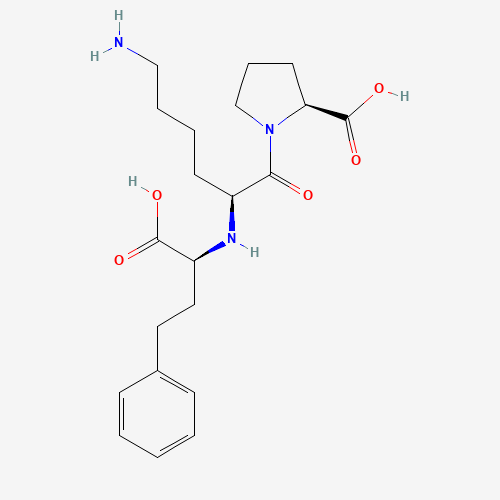
C21H31N3O5 |
| CAS Registry Number |
76547-98-3 |
| SMILES |
C1CC(N(C1)C(=O)C(CCCCN)NC(CCC2=CC=CC=C2)C(=O)O)C(=O)O |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
|
|
|

