| Pharmaceutical Information |
| Drug Name |
Fluciclovine f-18 |
| Drug ID |
BADD_D00910 |
| Description |
Fluciclovine is a [18F]-tagged synthetic analog of the amino acid L-leucine. It presents excellent diagnostic properties to be used in positron emission tomography (PET) imaging.[A31384] The structure of fluciclovine allows it to be uptaken by the tumoral cells by its amino acid transporter without incorporating in the metabolism within the body.[A31385] Fluciclovine was developed by Blue Earth Diagnostics, Ltd. and FDA approved in May 27, 2016.[L1049] |
| Indications and Usage |
Fluciclovine is indicated as a detection agent for positron emission tomography (PET) in men with suspected prostate cancer recurrence based on elevated blood prostate specific antigen (PSA) levels following prior treatment.[A31388] The overexpression of L-type amino acid transporters such as LAT1 and LAT3 that mediate the uptake of essential amino acids has been extensively reported as a tumoral mechanism of cell growth.[A31389] |
| Marketing Status |
approved |
| ATC Code |
V09IX12 |
| DrugBank ID |
DB13146
|
| KEGG ID |
D10860
|
| MeSH ID |
C117460
|
| PubChem ID |
450601
|
| TTD Drug ID |
D03QIP
|
| NDC Product Code |
69932-001 |
| UNII |
38R1Q0L1ZE
|
| Synonyms |
fluciclovine F-18 | (1R,3R)-1-amino-3(18F)fluorocyclobutane-1-carboxylic acid | cyclobutanecarboxylic acid, 1-amino-3-(fluoro-18F)-, trans- | F(18)-FACBC | F(18)1-amino-3-fluorocyclobutane-1-carboxylic acid | anti-(18F)FACBC | fluciclovine F 18 | fluciclovine (18F) | 1-amino-3-fluorocyclobutane-1-carboxylic acid | F-FACBC | Axumin | GE-148 (18F) | GE-148 F-18 | (18F)GE-148 | NMK 36 | NMK36 cpd | NMK-36 |
|
| Chemical Information |
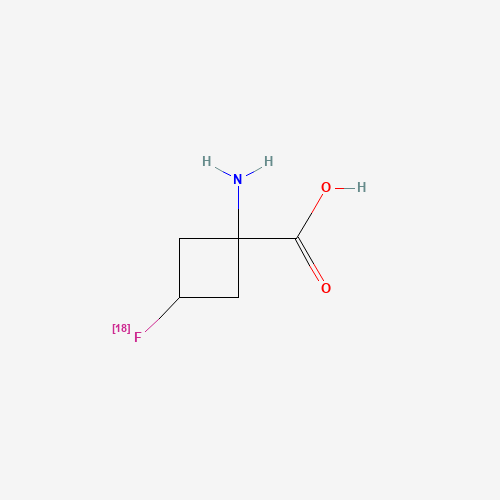
| Molecular Formula |
C5H8FNO2 |
| CAS Registry Number |
222727-39-1 |
| SMILES |
C1C(CC1(C(=O)O)N)F |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Dysgeusia | 07.14.03.001; 17.02.07.003 | - | - | | | Injection site erythema | 23.03.06.015; 12.07.03.001; 08.02.03.001 | - | - | Not Available | | Injection site pain | 12.07.03.011; 08.02.03.010 | - | - | Not Available |
|
The 1th Page
1
Total 1 Pages
|
|

