| Pharmaceutical Information |
| Drug Name |
Enzalutamide |
| Drug ID |
BADD_D00775 |
| Description |
Enzalutamide is an androgen receptor inhibitor for the treatment of castration-resistant prostate cancer. FDA approved on August 31, 2012. |
| Indications and Usage |
Enzalutamide is indicated for the treatment of patients with metastatic castration-resistant prostate cancer who have previously received docetaxel. |
| Marketing Status |
Prescription |
| ATC Code |
L02BB04 |
| DrugBank ID |
DB08899
|
| KEGG ID |
D10218
|
| MeSH ID |
C540278
|
| PubChem ID |
15951529
|
| TTD Drug ID |
D0QK5X
|
| NDC Product Code |
54893-0044; 55111-976; 0469-0625; 65392-2209; 68554-0106; 65015-876; 62128-0388; 65129-1376; 59285-018; 42385-733; 0469-0725; 11014-0003; 15308-1121; 0469-0125 |
| Synonyms |
enzalutamide | 4-(3-(4-cyano-3-(trifluoromethyl)phenyl)-5,5-dimethyl-4-oxo-2-thioxo-1-imidazolidinyl)-2-fluoro-N-methyl-benzamide | enzalutamide D3 | HC-1119 | HC 1119 | 4-(3-(4-cyano-3-(trifluoromethyl)phenyl)-5,5-dimethyl-4-oxo-2-thioxo-1-imidazolidinyl)-2-fluoro-N-(methyl-d3)benzamide | Xtandi | MDV 3100 | MDV3100 | MDV-3100 |
|
| Chemical Information |
| Molecular Formula |
C21H16F4N4O2S |
| CAS Registry Number |
915087-33-1 |
| SMILES |
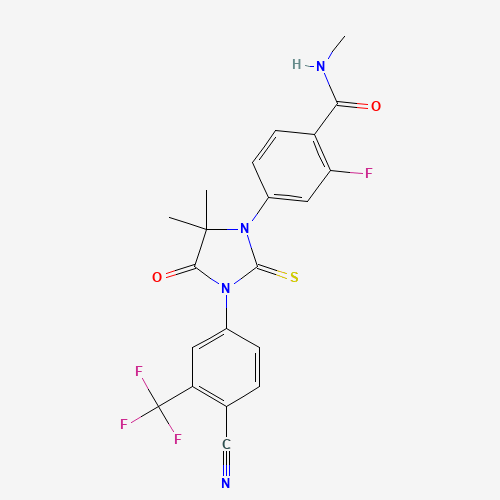
CC1(C(=O)N(C(=S)N1C2=CC(=C(C=C2)C(=O)NC)F)C3=CC(=C(C=C3)C#N)C(F)(F)F)C |
| Chemical Structure |
|
|
| ADR Related Proteins Induced by Drug |
| ADR Term |
Protein Name |
UniProt AC |
TTD Target ID |
PMID |
| Not Available | Not Available | Not Available | Not Available | Not Available |
|
| ADRs Induced by Drug |
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Spinal pain | 15.02.01.008; 17.10.01.020; 08.01.08.030 | 0.003677% | | Not Available | | Eye contusion | 06.11.01.011; 24.07.06.022; 12.01.04.024 | 0.002206% | | Not Available | | Tongue discomfort | 07.14.02.019 | 0.001471% | | Not Available |
|
|
|

