| Pharmaceutical Information |
| Drug Name |
Dexrazoxane hydrochloride |
| Drug ID |
BADD_D00634 |
| Description |
An antimitotic agent with immunosuppressive properties. Dexrazoxane, the (+)-enantiomorph of razoxane, provides cardioprotection against anthracycline toxicity. It appears to inhibit formation of a toxic iron-anthracycline complex. [PubChem]
The Food and Drug Administration has designated dexrazoxane as an orphan drug for use in the prevention or reduction in the incidence and severity of anthracycline-induced cardiomyopathy. |
| Indications and Usage |
For reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin hydrochloride dose of 300 mg/m^2 and would benefit from continued doxorubicin therapy. Also approved for the treatment of extravasation from intravenous anthracyclines. |
| Marketing Status |
approved; withdrawn |
| ATC Code |
V03AF02 |
| DrugBank ID |
DB00380
|
| KEGG ID |
D07807
|
| MeSH ID |
D064730
|
| PubChem ID |
6918223
|
| TTD Drug ID |
D07XVN
|
| NDC Product Code |
51991-942 |
| UNII |
5346058Q7S
|
| Synonyms |
Dexrazoxane | Razoxane, (S)-Isomer | Cardioxane | Cardioxan | Zinecard | ICRF-187 | ICRF 187 | ICRF187 | NSC-169780 | NSC 169780 | NSC169780 | ADR-529 | ADR 529 | ADR529 | Dexrazoxane Hydrochloride | Hydrochloride, Dexrazoxane | Razoxane, (S)-Isomer, Hydrochloride |
|
| Chemical Information |
| Molecular Formula |
C11H17ClN4O4 |
| CAS Registry Number |
149003-01-0 |
| SMILES |
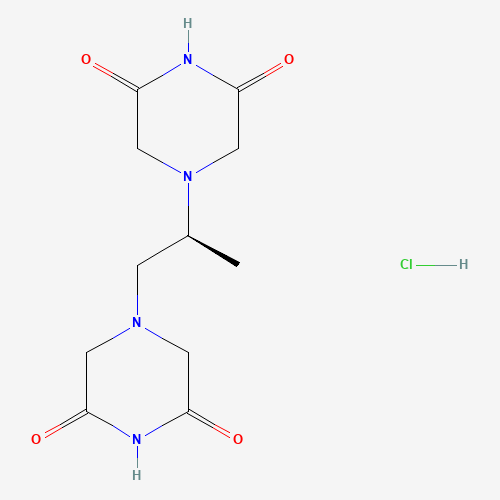
CC(CN1CC(=O)NC(=O)C1)N2CC(=O)NC(=O)C2.Cl |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Administration site streaking | 12.07.04.012; 08.02.04.012 | - | - | Not Available |
|
|
|

