| Pharmaceutical Information |
| Drug Name |
Dacarbazine |
| Drug ID |
BADD_D00568 |
| Description |
An antineoplastic agent. It has significant activity against melanomas. (from Martindale, The Extra Pharmacopoeia, 31st ed, p564). Dacarbazine with Oblimersen is in clinical trials for the treatment of malignant melanoma. |
| Indications and Usage |
For the treatment of metastatic malignant melanoma. In addition, dacarbazine is also indicated for Hodgkin's disease as a secondary-line therapy when used in combination with other antineoplastic agents. |
| Marketing Status |
approved; investigational |
| ATC Code |
L01AX04 |
| DrugBank ID |
DB00851
|
| KEGG ID |
D00288
|
| MeSH ID |
D003606
|
| PubChem ID |
2942
|
| TTD Drug ID |
D0Y7ZU
|
| NDC Product Code |
42973-149; 63323-128; 0143-9245; 0703-5075; 54875-0003; 63323-127 |
| UNII |
7GR28W0FJI
|
| Synonyms |
Dacarbazine | Dimethyl Imidazole Carboxamide | Carboxamide, Dimethyl Imidazole | Imidazole Carboxamide, Dimethyl | DTIC | ICDT | Decarbazine | 5-(3,3-Dimethyl-1-triazeno)imidazole-4-carboxamide | DIC | Dimethyl Triazeno Imidazole Carboxamide | NSC-45388 | NSC 45388 | NSC45388 | DTIC-Dome | DTIC Dome | DTICDome | Biocarbazine | Deticene |
|
| Chemical Information |
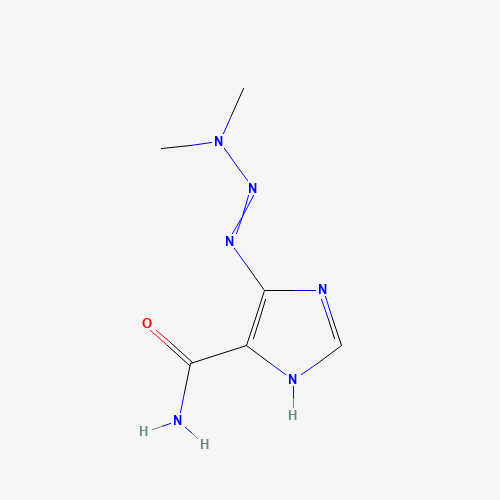
| Molecular Formula |
C6H10N6O |
| CAS Registry Number |
4342-03-4 |
| SMILES |
CN(C)N=NC1=C(NC=N1)C(=O)N |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Platelet toxicity | 12.03.01.057; 01.08.03.004 | 0.000280% | | Not Available | | Fixed eruption | 10.01.01.037; 08.01.06.025; 23.03.05.008 | - | - | Not Available |
|
|
|

