| Pharmaceutical Information |
| Drug Name |
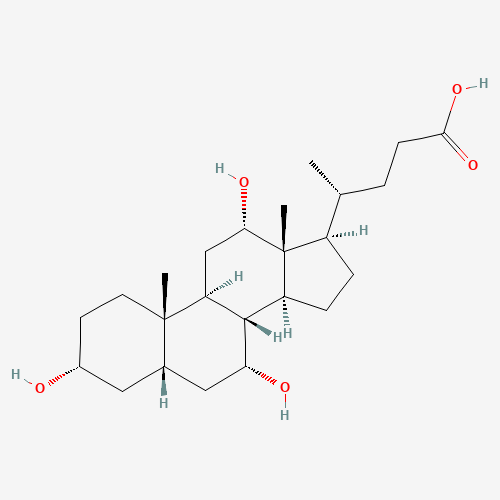
Cholic acid |
| Drug ID |
BADD_D00453 |
| Description |
A major primary bile acid produced in the liver and usually conjugated with glycine or taurine. It facilitates fat absorption and cholesterol excretion. [PubChem]
Cholic acid, formulated as Cholbam capsules, is approved by the United States Food and Drug Administration as a treatment for children and adults with bile acid synthesis disorders due to single enzyme defects, and for peroxisomal disorders (such as Zellweger syndrome). |
| Indications and Usage |
Oral cholic acid is indicated for: treatment of bile acid synthesis disorders due to single enzyme defects; and as adjunctive treatment of peroxisomal disorders including Zellweger spectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption. |
| Marketing Status |
approved |
| ATC Code |
A05AA03 |
| DrugBank ID |
DB02659
|
| KEGG ID |
D10699
|
| MeSH ID |
D019826
|
| PubChem ID |
221493
|
| TTD Drug ID |
D0OR2L
|
| NDC Product Code |
45043-002; 45043-001; 47049-002; 47049-001 |
| UNII |
G1JO7801AE
|
| Synonyms |
Cholic Acid | Cholbam |
|
| Chemical Information |
| Molecular Formula |
C24H40O5 |
| CAS Registry Number |
81-25-4 |
| SMILES |
CC(CCC(=O)O)C1CCC2C1(C(CC3C2C(CC4C3(CCC(C4)O)C)O)O)C |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
|

