| Pharmaceutical Information |
| Drug Name |
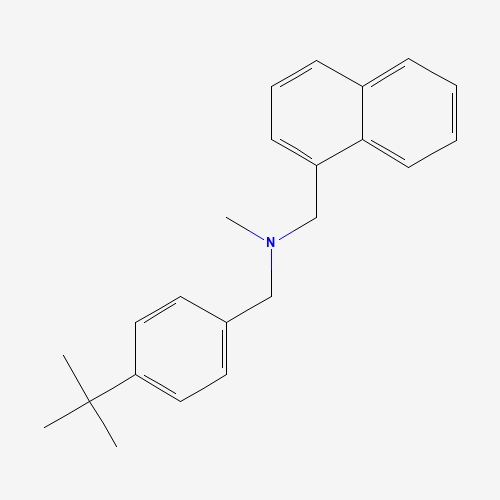
Butenafine |
| Drug ID |
BADD_D00321 |
| Description |
Butenafine hydrochloride is a synthetic benzylamine antifungal agent. Butenafine's mechanism of action is believed to involve the synthesis inhibition of sterols. In particular, butenafine acts to inhibit the activity of the squalene epoxidase enzyme that is essential in the formation of sterols necessary for fungal cell membranes. |
| Indications and Usage |
For the topical treatment of the following dermatologic infections: tinea (pityriasis) versicolor due to M. furfur, interdigital tinea pedis (athlete’s foot), tinea corporis (ringworm) and tinea cruris (jock itch) due to E. floccosum, T. mentagrophytes, T. rubrum, and T. tonsurans. |
| Marketing Status |
approved |
| ATC Code |
D01AE23 |
| DrugBank ID |
DB01091
|
| KEGG ID |
D07596
|
| MeSH ID |
C067594
|
| PubChem ID |
2484
|
| TTD Drug ID |
D0B4DC
|
| NDC Product Code |
Not Available |
| UNII |
91Y494NL0X
|
| Synonyms |
butenafine | Mentax | butenafine hydrochloride | N-4-tert-butylbenzyl-N-methyl-1-naphthalene methylamine hydrochloride | KP 363 | KP-363 |
|
| Chemical Information |
| Molecular Formula |
C23H27N |
| CAS Registry Number |
101828-21-1 |
| SMILES |
CC(C)(C)C1=CC=C(C=C1)CN(C)CC2=CC=CC3=CC=CC=C32 |
| Chemical Structure |
|
|
| ADRs Induced by Drug |
|
|
*The priority for ADR severity classification is based on FAERS assessment, followed by the most severe level in CTCAE rating. If neither is available, it will be displayed as 'Not available'.
**The 'Not Available' level is hidden by default and can be restored by clicking on the legend twice..
|
|
| ADR Term |
ADReCS ID |
ADR Frequency (FAERS)
|
ADR Severity Grade (FAERS)
|
ADR Severity Grade (CTCAE)
|
| Dermatitis contact | 12.03.01.040; 10.01.01.003; 23.03.04.004 | - | - | Not Available | | Dysgeusia | 17.02.07.003; 07.14.03.001 | - | - | | | Erythema | 23.03.06.001 | - | - | Not Available | | Pain | 08.01.08.004 | - | - | | | Pruritus | 23.03.12.001 | - | - | |
|
The 1th Page
1
Total 1 Pages
|
|

